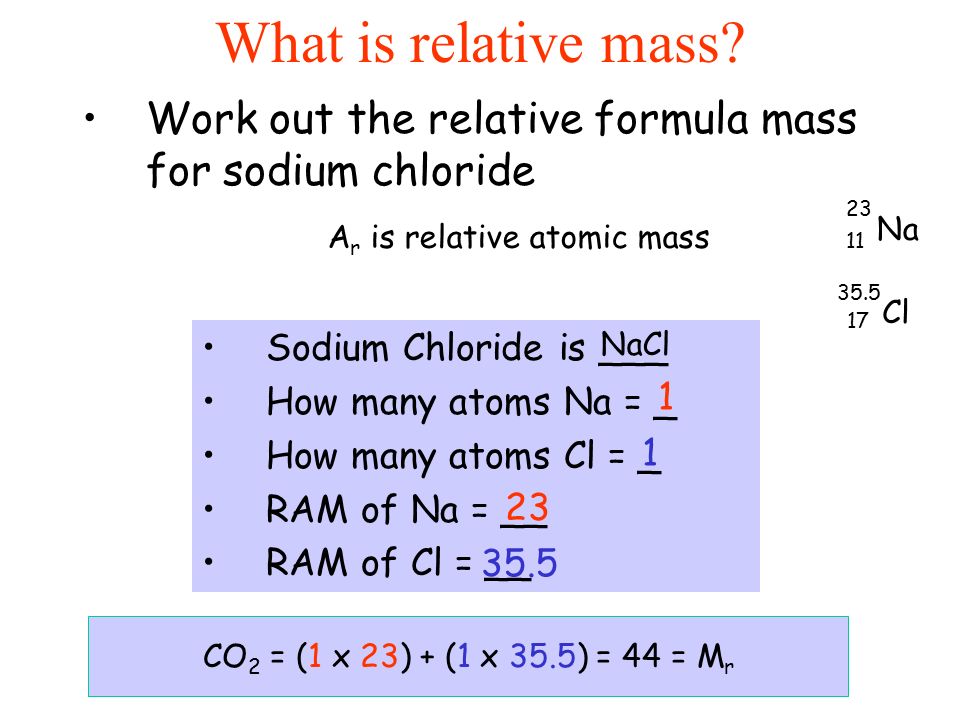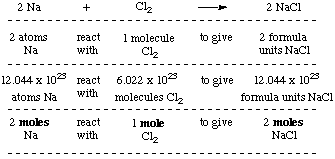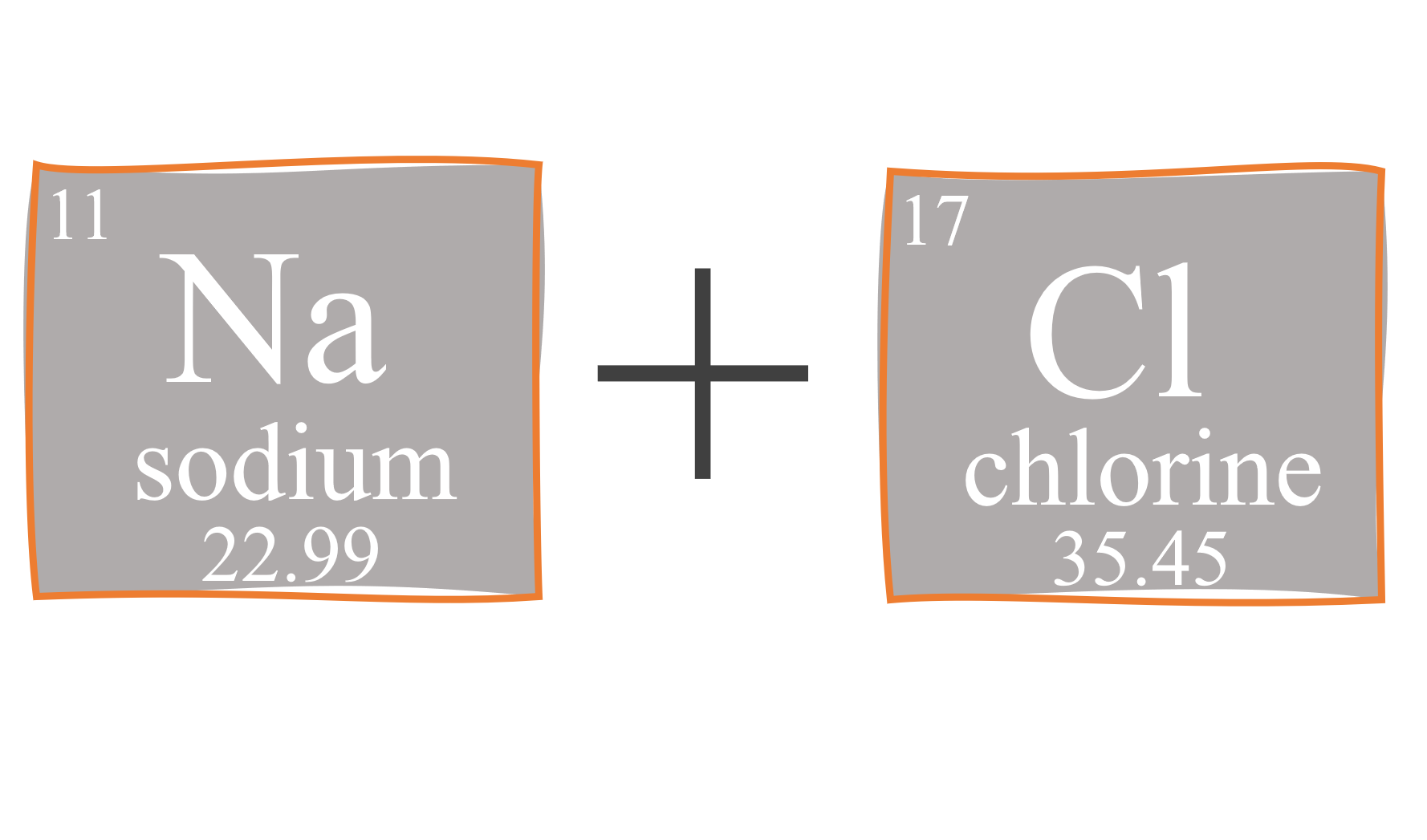
MOLAR MASS Molar mass of a substance = mass in grams of one mole of the substance. A compound's molar mass is NUMERICALLY equal to its formula mass. Formula. - ppt download

Molar Mass. A compound is a collection of bonded together. Ethanol, CH 3 CH 2 OH, is made up of : carbon atoms, hydrogen atoms, and oxygen atom, ppt download

Densities of phonon states of a crystal of the NaCl structure for the... | Download Scientific Diagram

25. The molecular weight of NaCl determined by studying freezing point depression of its 0.5% aqueous solution is 30. The apparent degree of dissociation of NaCl is (1) 0.95 (2) 0.45 (3) 0.60 (4) 0.35

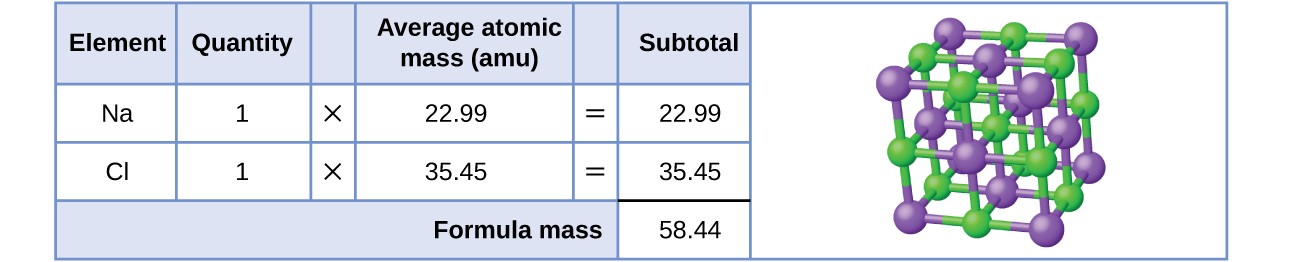
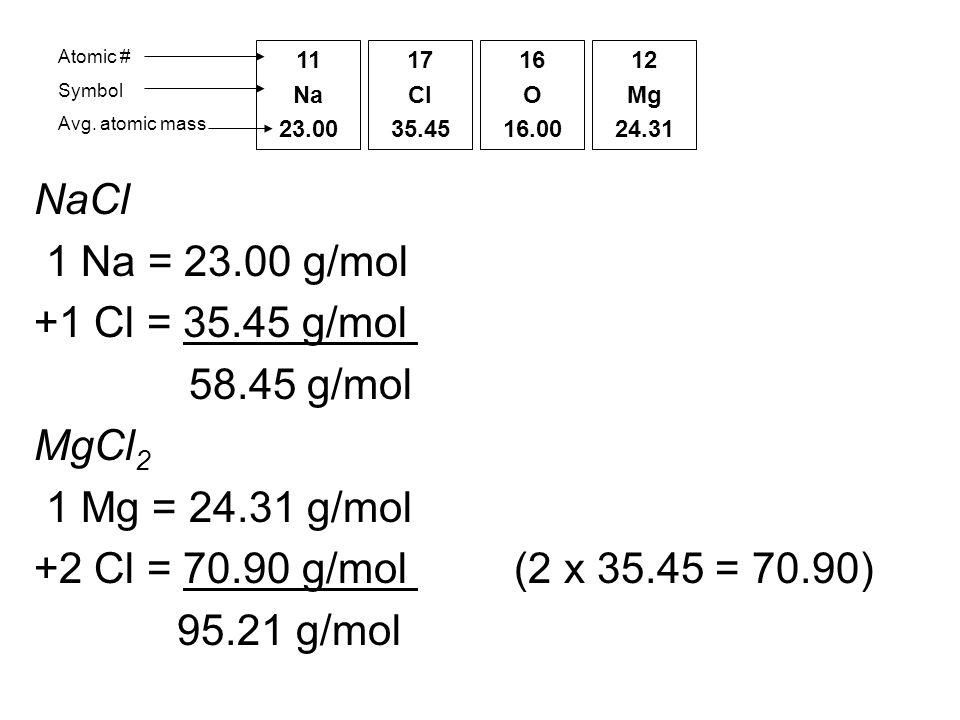
1 Formula mass is the sum of the atomic masses (in amu) in a formula unit of an ionic compound. 1Na22.99 amu 1Cl amu NaCl amu For any ionic. - ppt download