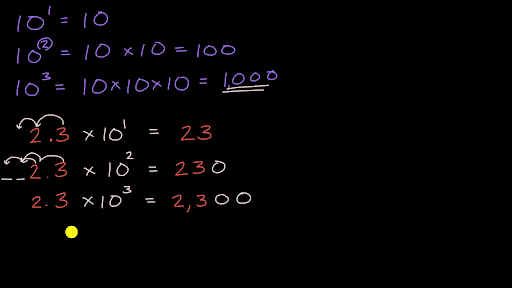At ntp what will be volume of molecules of 6 022 10 raise to power 23 of h2 Q3 - Chemistry - Some Basic Concepts of Chemistry - 13715971 | Meritnation.com
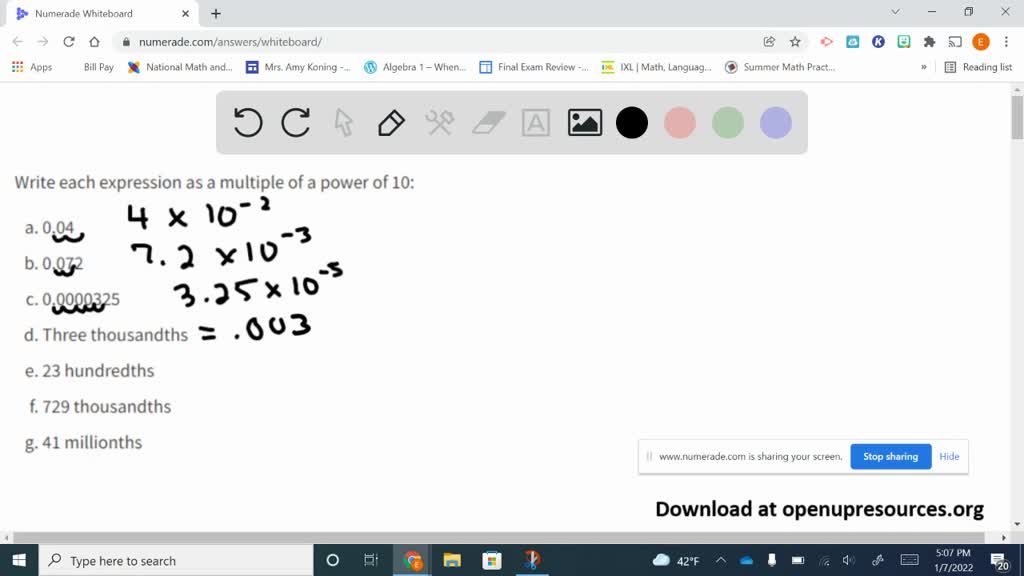
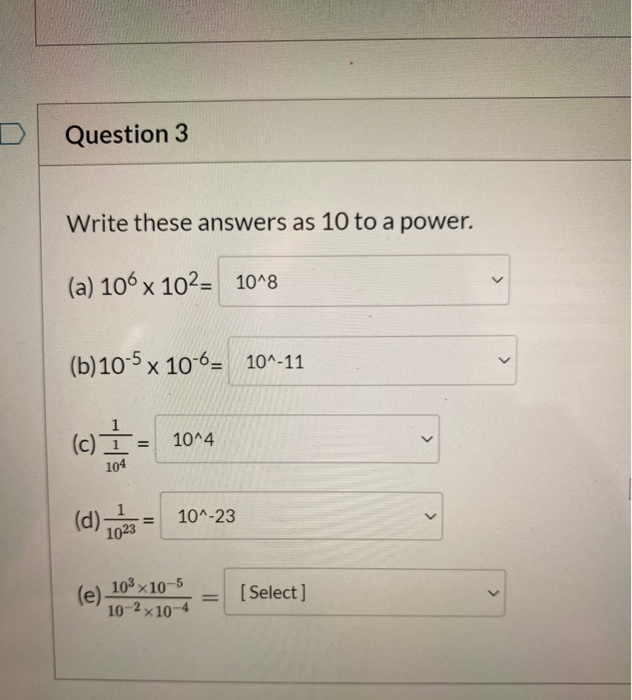
SOLVED:Write each expression as a multiple of a power of 10: a. 0.04 b. 0.072 c. 0.0000325 d. Three thousandths e. 23 hundredths f. 729 thousandths g. 41 millionths
Calculate the number of moles present in: 3.011 x 10^23 number of oxygen atoms. 60 g of calcium - Sarthaks eConnect | Largest Online Education Community

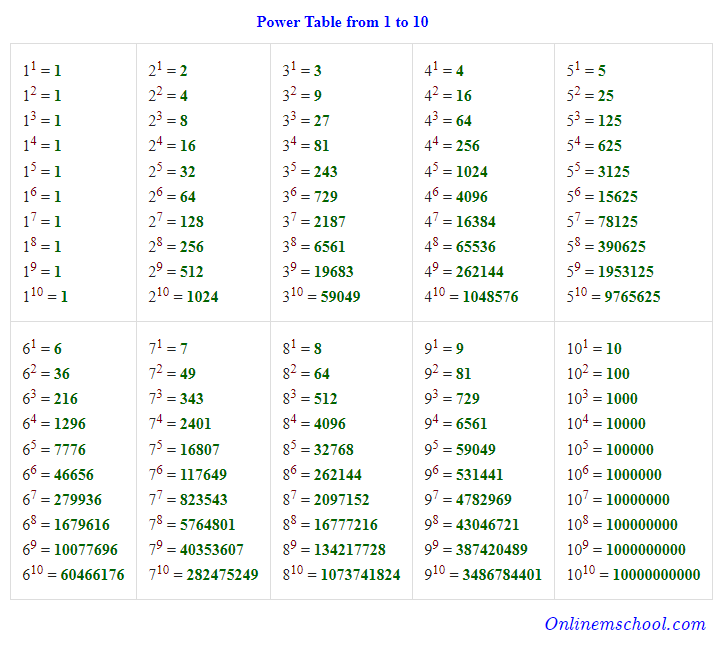
SCIENTIFIC NOTATION A value written as the product of two numbers: a coefficient and 10 raised to a power. Ex: 602,000,000,000,000,000,000,000 is ppt download